You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
In implant-supported reconstruction cases, gingival esthetics is influenced by many factors throughout the treatment process. In an interdisciplinary team treatment approach, effective and clear communication between the surgical team member, restoring dentist, and laboratory technician are essential to achieving the desired treatment outcome. Below, the authors discuss four principle areas of treatment that are used in the surgical and restorative phases of treatment.
Managing the Hard Tissues
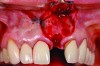
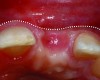
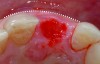
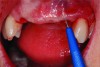
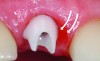
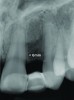
It is uncommon to find thick bone walls forming a socket in the esthetic zone, as seen in Figure 1. The buccal plate thickness in a socket is, on average, 0.9 mm +/- 0.4 mm at the mid-root area and roots are often positioned buccally in the ridge.1 As a result, ridge resorption after extraction is common and can be even more significant when the walls of the socket are affected by dehiscences or fenestrations (Figure 2). Although facial ridge resorption is more common, some degree of vertical resorption is also expected after an extraction; both become more significant when more than one tooth is extracted.2-4 Ridge preservation procedures can minimize the degree of post-extraction ridge remodeling through the addition of a variety of bone graft materials at the time of extraction. Figure 3 clearly illustrates moderate buccal ridge resorption that develops post-extraction, while Figure 4 shows little remodeling as a result of placement of an allograft at the time of extraction.
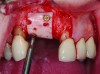
Where ridge preservation procedures are not possible or where the ridge is compromised by disease, ridge augmentation procedures are often required in the implant treatment plan, in order to allow restoratively driven implant placement. Autogenous bone grafts have been recognized as the material of choice, but many clinicians are increasingly using alternative materials to avoid a second surgical site and the morbidity that may be associated with procurement of the graft. Bone allografts in particulate form have long been used in periodontal therapy and in ridge augmentation for implant site development. Although not extensively researched, bone allografts in block form are now being used as alternatives to autogenous block grafts in the augmentation of more significant ridge defects.5-8 In Figure 5, a contoured allogenic corticocancellous block graft was stabilized with fixation screws, surrounded by particulate bone in the proximal voids and protected with a cross-linked collagen membrane. This case illustrates the similarity to conventional autogenous block graft procedures. A retrospective review of the results of the first 10 cases using this material alternative is promising, with bone volume gains similar to that achieved with autografts. Bone volume and its post-restoration stability can be influenced by implant design, with literature support for platform switching or micro-grooved implant collars.9-12 It is well-documented that 3-dimensionally correct implant placement is important, because this impacts bone stability in the postsurgical period.13
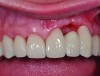
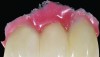
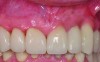
Although there are many approaches and materials that may be used to augment a ridge, there are treatment limitations in the augmentation of vertically deficient ridges, especially those where bone levels on adjacent teeth have been adversely affected by disease processes or surgical complications. Gingival masks or “prosthetic” gingival tissues sometimes become necessary to hide these deficiencies. As illustrated in Figure 6 through Figure 8, the current trend to use gingival-colored composites, which can be “refreshed or replaced,” appears promising from an esthetic perspective, but is at present viewed as heroic and of questionable long-term predictability.
The Gingival Biotype
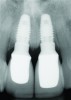
Most of the literature on implant success rates has not identified a correlation with the gingival biotype, although it is increasingly accepted that the biotype and tissue volume have an important impact on the esthetic outcome and minimizing the risk for post-restoration tissue instability. In Figure 9 and Figure 10, adjacent implants placed 10 years previously are successful in terms of integration, but negative soft-tissue changes—both facial and inter-implant—have resulted in poor esthetics due to recession and soft-tissue color changes from titanium shine-through.14,15 Tissue color is less impacted by transmucosal restorative materials when greater than 3 mm thick, but this thickness is rarely seen in the transmucosal area, even in grafted cases.16,17
Microsurgical techniques used in root coverage procedures have been shown to produce results superior to conventional macrosurgery.18 The impact of connective tissue grafting on peri-implant tissue volume may be viewed as a critical tool to the restoring dentist from the viewpoint of tissue color and stability (Figure 11 and Figure 12). By improving the tissue volume, this also gives the restoring clinician the possible ability to manipulate and define the gingival tissues with provisional restorations. Durable temporary materials that can be modified chairside are used to convert ridges with indistinct anatomy to ones with sculpted form, simulating papilla as well as crisp facial and palatal tissues. This aspect of treatment was previously reviewed in detail in an article by the authors.19
Surgical and Restorative Strategies
Surgical techniques, such as the implant placement protocol and the restoration protocol relative to the time of surgery, may also impact the soft-tissue form and its stability. With current treatment-planning tools—such as software used to virtually plan implant procedures coupled with 3-dimensionally accurate surgical guides—the clinician can anticipate the surgical and restoration approach that can be undertaken, thus decreasing the likelihood of surprises that impact a treatment plan during the surgical phase of treatment. Importantly, this subsequently minimizes the challenges that will be encountered by the restoring dentist. Using cone-beam computed tomography (CBCT) and a guided software planning system, virtual implant placement in the left central incisor area shown indicated that bone grafting would be required to fulfill the ridge volume requirements on the facial of the implant (Figure 13 and Figure 14). In this case, an implant design offering high primary stability was selected and facial bone grafting was completed to increase the facial bone volume to at least 2 mm. Figure 15 illustrates the implant site preparation using a surgical guide and flap access to permit facial bone grafting. When initial implant stability is adequate, implants may be restored at the time of placement or shortly thereafter, as illustrated in Figure 16. The potential merits of this are based on the patient’s perceived benefits, but from the clinician’s perspective, the potential advantages include coordination of the integration period and the soft-tissue architecture development. The prospective clinical trial of Block and coauthors found that immediate implant restoration had a positive impact on facial tissue levels, with better facial tissue positions compared with cases that underwent delayed restoration.20
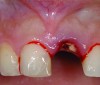
Similarly, the approach of immediate implant placement and restoration is increasingly being used by clinicians, although the esthetic risks are considered high by some, because the procedure requires precision and careful management of hard and soft tissues. Aside from the patient-driven rationale for this approach, in appropriately selected cases, tissue guidance by the fixed restoration may lead to enhanced papilla development and better post-restoration facial tissue stability, because the soft tissues are less likely to slump or lose their anatomy. De Rouck and coauthors reported enhanced results with immediate versus delayed implant restoration.21 The case illustrated in Figure 17 involved extraction of tooth No. 9 and immediate implant placement, placement of a xenograft material in the residual horizontal defect, and fabrication/delivery of a provisional restoration. Implant placement at the time of tooth extraction may not prevent ridge resorption from occurring, but management of the residual horizontal defect between the socket wall and implant surface can influence the tissue level, particularly on the facial aspect.22 It is the authors’ observation that specific treatment with an anorganic bone graft (xenograft) can reduce buccal ridge resorption, and as a consequence, will positively influence the facial soft-tissue levels.
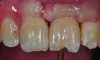
After a 3-month integration period, removal of the provisional restoration shows well-developed tissue surrounding the implant collar (Figure 18). The adjacent tooth was prepared and impressed for a porcelain restoration, and an impression coping was customized to record the position of the implant and its soft-tissue anatomy (Figure 19). Zirconia is increasingly recognized as an abutment material with many benefits in the esthetic zone, but its success is clearly linked to basic decisions in the surgical phase of treatment. An ideally designed zirconia abutment mimics the preparation of the adjacent No. 8, with minimal submergence of the margin on the implant and natural tooth (Figure 20); the ideal restorative result demands a pleasing balance of white and pink.
Hard and Soft-Tissue Structure
Just as a root and its position affect the soft-tissue contours and levels in the natural dentition, abutment contour in the transmucosal area can have an important impact on gingival form. Designing abutment contours to fully support and sculpt tissues in the sulcus area was common in the past; however, undercontouring the abutment as it leaves the head of the implant and moves toward the free gingival margin may offer the advantage of producing a thicker gingival collar, which may act as a protective barrier for the underlying bone (Figure 21). The illustrative case reveals a thickened circumferential band of tissue in the transmucosal area. In effect, the crown at the margin level and the coronal 0.5 mm to 1 mm of the abutment effectively support the contour of the marginal gingival tissue. Communication with the laboratory is of paramount importance if this simple step is considered in the prosthetic phase of treatment, allowing the dental technician to incorporate this design feature into the abutment using CAD/CAM technology.
When zirconia is used as an abutment material, it offers the clinician numerous advantages, including high biocompatibility. However, biocompatibility can be affected by the handling of the material. The work of Mustafa et al and Lee et al both indicate that abutment modification can impact how the soft tissue will respond. Milled zirconia surfaces were shown to have significantly higher percentages of cell attachment and proliferation than polished zirconia surfaces.23,24
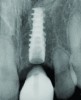
Importantly, the pleasing color of zirconia, whether it is white or colored, provides the opportunity to position the cement line more coronally. Titanium abutments can cast a shadow through thin peri-implant tissues. This often results in the clinician’s pushing the cement line considerably deeper in an effort to hide the titanium’s “graying” effect.25,26 Several examples of cement trapping illustrated clinically and radiographically were used to support this important problem in the restoration of implants (Figure 22).27 The consequences of cement trapping are variable; they range from minor symptoms of tissue discomfort or sensitivity, to clinical signs of inflammation characteristic of peri-implant mucositis, and, most advanced, bone loss or peri-implantitis (Figure 23). In an unpublished study by the authors, 55 restorations and their abutments were removed as a unit by accessing the abutment screws through the occlusal or incisal. These restorations were removed for the following reasons: they were deemed to be esthetically unsatisfactory by the patient and/or dentist; they had porcelain chipping; or they were incompletely cemented. Retained cement was recorded on 29 of the 55 units removed. This problem appeared most frequently when the depth of placement of the cement line was significant relative to the gingival margin (more than 2 mm). Although zirconia offers a color advantage, it is still important to emphasize that the technician must design the abutment in reference to the tissue topography. Figure 24 illustrates an abutment that has not been properly designed in the interproximal area with respect to the soft-tissue profile, while Figure 20 previously illustrated an appropriately designed abutment avoiding these deeply positioned marginal areas. When coupled with a suitable cementation technique, such as using a deflective cord below the cement line, these steps can minimize the introduction of cement into the sulcular area.
As discussed, new implant designs can influence bone and, therefore, soft-tissue levels and stability along with several other important treatment-planning considerations. Similarly, selection of the correct implant size in an area of treatment also impacts tissue levels around the implant and the adjacent teeth. The use of a small-diameter implant (3 mm) of the platform-switching concept was used in a space of limited mesiodistal dimension. Excellent bone levels were noted around the implant, and importantly, with no adverse effect on marginal bone levels on the adjacent teeth (Figure 25 and Figure 26). If a wider implant diameter had been selected, the periodontium of the adjacent teeth may have been compromised had any remodeling of the bone around the implant occurred.
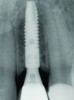
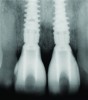
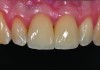
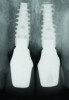
In the final case, adjacent implants were placed in healed sites No. 8 and No. 9 and provisionalized at the time of surgery (Figure 27 and Figure 28). This case illustrates the effect of platform switching on bone and gingival tissue stability, as well as recognizing that appropriate diameter and spacing is important to maintain ideal ridge contour. The gingival tissue anatomy between implants is characteristically shorter than papilla form between adjacent teeth, and, as a consequence, it is important to incorporate implant designs and procedures that minimize bone-level changes and associated soft-tissue recession. The depth of implant placement was subcrestal relative to the ridge crest, based on the future desired free gingival tissue level, while allowing prosthetic running room. The studies of Barros et al and Veis et al noted that implants positioned subcrestally (as seen in Figure 29) showed better crestal bone preservation than those placed equicrestally or supracrestally.28,29 The implant design used in this case achieves excellent primary stability. Along with the optimal 3-dimensional positioning of the implants and, specifically, the subcrestal placement, as well as the immediate provisionalization protocol, these concepts and implants have contributed to a pleasing esthetic outcome, as seen in Figure 30.
Conclusion
The treatment concepts and procedures illustrated in this review are presented in comprehensive chapters on treatment planning for implant esthetics in two publications by the authors.30,31 The clinical cases presented illustrate that gingival esthetics is influenced by many factors throughout the treatment process, and that communication between the surgical team member, restoring dentist, and laboratory technician are essential to achieving the desired treatment outcome.
Acknowledgments
The authors would like to thank their ceramists, Aki Miyazaki, Klaas van der Walt, and Nico Pienaar.
About the Authors
Sonia Leziy, DDS
Private Practice, Imperio Group Dental Health Specialists
North Vancouver, British Columbia, Canada
Brahm Miller, DDS
Private Practice, Imperio Group Dental Health Specialists
North Vancouver, British Columbia, Canada
References
1. Lau SL, Chow J, Li W, Chow LK. Classification of maxillary central incisors—implications for immediate implant in the esthetic zone. J Oral Maxillofac Surg. 2011;69(1):142-153.
2. Schropp L, Wenzel A, Kostopoulos L, Karring T. Bone healing and soft tissue contour changes following single tooth extraction: A clinical and radiographic 12-month prospective study. Int J Periodontics Restorative Dent. 2003;23(4):313-323.
3. Araújo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32(2):212-218.
4. Nevins M, Camelo M, De Paoli S, et al. A study of the fate of the buccal wall of extraction sockets of teeth with prominent roots. Int J Periodontics Restorative Dent. 2006;26(1):19-29.
5. Lyford RH , Mills MP , Knapp CI, et al. Clinical evaluation of freeze-dried block allografts for alveolar ridge augmentation: a case series. Int J Periodontics Restorative Dent. 2003;23(5):417-425.
6. Nissan J, Mardinger O, Calderon S, et al. Cancellous bone block allografts for augmentation of the anterior atrophic maxilla. Clin Implant Dent Relat Res. 2009;13(2):104-111.
7. Wallace S, Gellin R. Clinical evaluation of freeze-dried cancellous block allografts for ridge augmentation and implant placement in the maxilla. Implant Dent. 2010;19(4):272-279.
8. Waasdorp J, Reynolds MA. Allogenic bone only grafts for alveolar ridge augmentation: a systematic review. Int J Oral Maxillofac Implants. 2010;25(3):525-531.
9. Gastaldo JF, Cury PR, Sendyk WR. Effect of the vertical and horizontal distances between adjacent implants and between a tooth and an implant on the incidence of interproximal papilla. J Periodontal. 2004;75(9):1242-1246.
10. Rodríguez-Ciurana X, Vela-Nebot X, Segalà-Torres M, et al. The effect of interimplant distance on the height of the interimplant bone crest when using platform-switched implants. Int J Periodontics Restorative Dent. 2009;29(2):141-151.
11. Becker J, Ferrari D, Herten M, et al. Influence of platform switching on crestal bone changes at non-submerged titanium implants: a histomorphometrical study in dogs. J Clin Periodontol. 2007;34(12):1089-1096.
12. Shapoff CA, Lahey B, Wasserlauf PA , Kim DM. Radiographic analysis of crestal bone levels around Laser-Lok collar dental implants. Int J Periodontics Restorative Dent. 2010;30(2):129-137.
13. Grunder U, Gracis S, Capelli M. Influence of the 3-D bone-to-implant relationship on esthetics. Int J Periodontics Restorative Dent. 2005;25(2):113-119.
14. Linkevicius T, Apse P, Grybauskas S, Pulsys A. The influence of soft tissue thickness on crestal bone changes around implants: a 1-year prospective controlled clinical trial. Int J Oral Maxillofac Implants. 2009;24(4):712-719.
15. Lee A, Fu JH, Wang HL. Soft tissue biotype affects implant success. Implant Dent. 2011;20(3);e38-47.
16. Jung RE, Sailer I, Hämmerle CH, et al. In vitro color changes of soft tissues caused by restorative materials. Int J Periodontics Restorative Dent. 2007;27(3):251-257.
17. Zembic A, Sailer I, Jung RE, Hämmerle CH. Randomized-controlled clinical trial of customized zirconia and titanium implant abutments for single-tooth implants in canine and posterior regions: 3-year results. Clin Oral Implants Res. 2009;20(8):802-808.
18. Burkhardt R, Lang NP. Coverage of localized gingival recessions: comparison of micro- and macrosurgical techniques. J Clin Periodontol. 2005;32(3):287-293.
19. Leziy SS, Miller BA. Developing ideal implant tissue architecture and pontic site form. Quintessence of Dent Technol. 2007;30(1):143-154.
20. Block MS, Mercante DE, Lirette D, et al. Prospective evaluation of immediate and delayed provisional single tooth restorations. J Oral Maxillofac Surg. 2009;67(11 Suppl):89-107.
21. De Rouck T, Collys K, Wyn I, Cosyn J. Instant provisionalization of immediate single-tooth implants is essential to optimize esthetic treatment outcomes. Clin Oral Implants Res. 2009;20(6):566-570.
22. Chen ST , Darby IB, Reynolds EC. A prospective clinical study of nonsubmerged immediate implants: clinical outcomes and esthetic results. Clin Oral Impl Res. 2007;18(5):552-562.
23. Mustafa K, Wennerberg A, Arvidson K, et al. Influence of modifying and veneering the surface of ceramic abutments on cellular attachment and proliferation. Clin Oral Impl Res. 2009;19(11):1178-1187.
24. Lee SW, Kim SY , Rhyu IC, et al. Influence of microgroove dimension on cell behavior of human gingival fibroblasts cultured on titanium substrata. Clin Oral Impl Res. 2009;20(1):56-66.
25. Watkin A, Kerstein RB. Improving darkened anterior peri-implant tissue color with zirconia custom implant abutments. Compend Contin Educ Dent. 2008;29(4):238-240, 242.
26. Koutayas SO , Vagkopoulou T, Pelekanos S, et al. Zirconia in dentistry: part 2. Evidence-based clinical breakthrough. Eur J Esthet Dent. 2009;4(4):348-380.
27. Wilson TG Jr. The positive relationship between excess cement and peri-implant disease: a prospective clinical endoscopic study. J Periodontol. 2009;80(9):1399-1392.
28. Barros RR, Novaes AB Jr, Muglia VA, et al. Influence of interimplant distances and placement depth on peri-implant remodeling of adjacent and immediately loaded Morse cone connection implants: a histomorphometric study in dogs. Clin Oral Implants Res. 2010;21(4):371-378.
29. Veis A, Parissis N, Tsirlis A, et al. Evaluation of peri-implant marginal bone loss using modified abutment connections at various crestal level placements. Int J Periodontics Restorative Dent. 2010;30(6):609-617.
30. Leziy S, Miller B. Esthetics in implant therapy: a blueprint for success. In: Cohen M, ed. Interdisciplinary Treatment Planning: Principles, Design, Implementation. Hanover Park, IL: Quintessence Publishing Co, Inc; 2008: chap 3.
31. Leziy S, Miller B. The papilla between adjacent implants: Treatment planning to optimize aesthetic outcomes. Hanover Park, IL: Quintessence Publishing Co, Inc; Fall 2011: chapter in press.